Oral tumors account for around 6% of all canine tumors, including both benign and malignant tumors. The most common oral tumors in dogs include: melanoma, squamous cell carcinoma, sarcomas, and ameloblastomas. The initial treatment of oral tumors is often similar. However, the definitive treatment options and prognosis is highly variable based on the individual tumor type, as well as its stage.
In a large number of cases, tumors are identified incidentally during annual dental exams. It is important to evaluate a patient’s whole mouth, including base of the tongue and tonsils during any dental procedure or other anesthetic event requiring intubation. Potential clinical signs owners may report include halitosis, bleeding from the mouth, hesitancy eating hard kibble, or the appearance of a visible mass.
The most recognized staging system for oral tumors utilizes the World Health Organization Tumor Node Metastasis (TNM) system. The stage reflects an assessment of the primary tumor’s size, the regional lymph nodes (fixed, mobile, or metastatic), along with the presence of distant metastasis. Larger tumors, even without the presence of local or distant metastasis, can be of higher stage. The specific tumor type provides greater detailof the risk of metastasis to either the regional lymph nodes and/or distant sites. Specific recommendations for further testing is outlined below. In addition to the assessment of lymph nodes and lungs, full lab work should be considered. This information may guide further recommendations for both staging and treatment.
Staging
The extent of staging varies based on the individual tumor type. Oral melanoma is both locally aggressive and highly metastatic at the time of diagnosis. In contrast, oral squamous cell carcinomas have a lower metastatic potential and lower risk at the time of diagnosis. Fibrosarcomas tend to be extensive and aggressive locally, but have a low metastatic potential. The recommended initial staging for oral tumors includes regional lymph node evaluation and thoracic radiographs (as recommended above).
The size of regional lymph nodes has been shown to be of limited utility in the detection of metastasis. One study evaluating regional lymph node metastasis in dogs with oral melanoma found 40% of dogs with normal size lymph nodes had metastatic disease.1 In addition, lymphatic drainage is highly variable around the head and neck of dogs. Roughly 60% of dogs reportedly have contralateral metastasis compared to ~90% of dogs that develop ipsilateral metastasis.2. In addition, disease can be found within the medial retropharyngeal lymph nodes without first affecting the mandibular lymph nodes.3 The risk of pulmonary metastasis at the time of diagnosis is highest in oral melanoma, however has been noted in sarcomas and squamous cell carcinoma. There is no metastatic potential associated with ameloblastomas.
Diagnosing Oral Tumors in Dogs
The diagnosis of canine oral tumors may require multiple steps. Cytology or fine needle aspirate may provide a diagnosis in some tumor types, such as squamous cell carcinoma. However, in other tumor types (ie. melanoma), cytology may be unrewarding. Melanomas can appear cytologically similar to round cells, epithelial cells, or mesenchymal cells. Oral cavity sarcomas often do not exfoliate well,limiting the diagnostic benefit of cytology. More commonly, a biopsy is necessary to provide a definitive diagnosis. Melanomas remain elusive and can be a diagnostic challenge due to cell shape and the possibility of the tumor being amelanotic. It is often necessary to consider immunohistochemistry (IHC) to provide the definitive diagnosis in oral melanoma.4
Treatment Options
Surgery
The primary treatment recommendation for oral tumors in dogs is surgical resection. This surgery may entail extensive procedures, including mandibulectomy, maxillectomy, or glossectomy. The goal with any surgery to remove an oral tumor, including ameloblastomas, is wide margins to limit the potential for local recurrence. Surgery is the primary modality for fibrosarcomas, SCC, and ameloblastomas. Before considering aggressive surgery, complete staging and advanced imaging should be completed. A CT scan will often reveal extensive disease beyond what can be appreciated both grossly and radiographically. In addition, advanced imaging can be of further utility in assessing all regional lymph nodes.
Radiation Therapy
Radiation therapy can be considered in the primary local treatment and/oradjunctive treatment setting. Palliative radiation can be utilized in OMM if the primary tumor is not amenable to surgical excision or other negative prognostic factors are present. Melanoma is responsive to higher dose-per-fraction (coarse fraction) treatments, as utilized in palliative protocols. The reported overall response rate is >75%, with nearly 2/3 of cases experiencing complete responses. In dogs without negative prognostic factors, the median survival time reported is 21 months.5 Radiation therapy is more often considered following surgery in the management of oral squamous cell carcinoma. Sarcomas are considered resistant to radiation therapy. However, radiation therapy may be considered following incomplete resection of sarcomasor in the palliative setting when surgery is not an option. Ameloblastomas are responsive to radiation therapy with a reported prognosis of 37 months.6
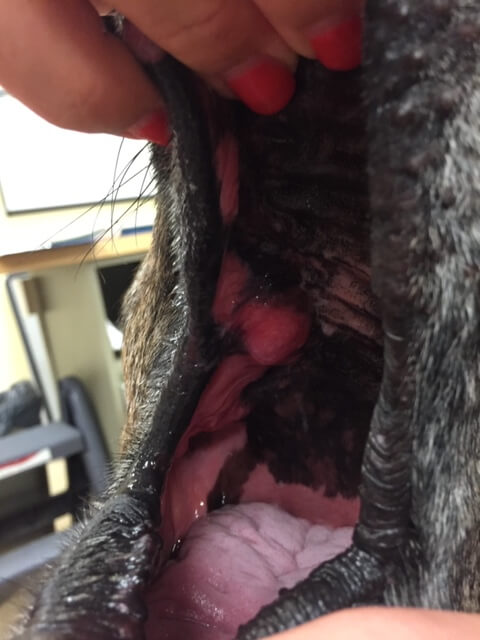
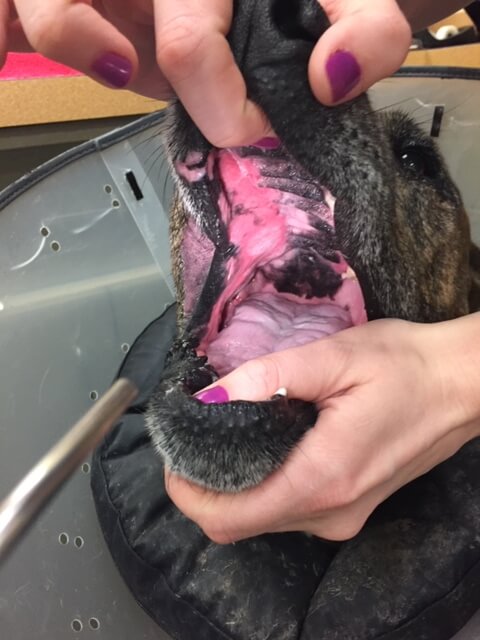
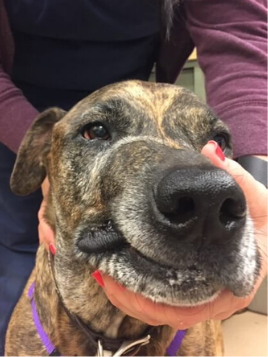
Canine oral FSA, treated with surgery and radiation.
Systemic Therapy
The role of systemic therapy in the management of canine oral tumors is varied. Melanoma tends to be poorly responsive to traditional systemic therapy such as chemotherapy. The published options include carboplatin with a response rate of 28%7 and cisplatin with a response rate of 18%.8 SCC has been shown to be responsive to a number of chemotherapies, including bleomycin in combination with electrochemotherapy, carboplatin, Palladia, and Piroxicam alone. Sarcomas are poorly responsive in the measurable disease setting to systemic therapy. Although the metastatic potential is reportedly decreased with osteosarcoma of the oral cavity compared to the appendicular skeleton, chemotherapy is often still recommended. Lastly, ameloblastomas are responsive to intralesional bleomycin with a reported rate of complete responses in 85% of cases. Local recurrence was not reported for >800 days.9,10
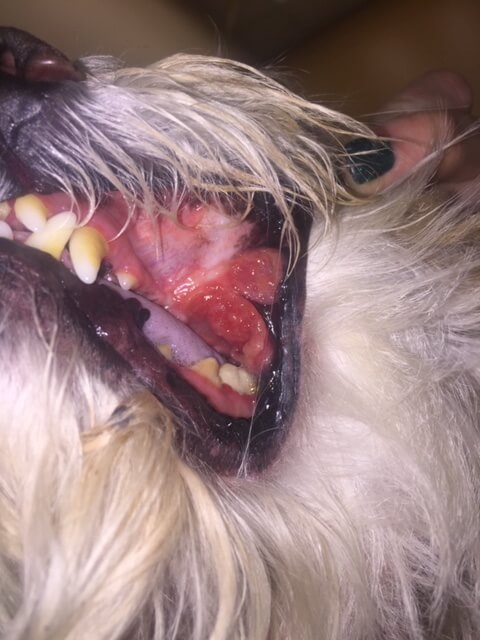
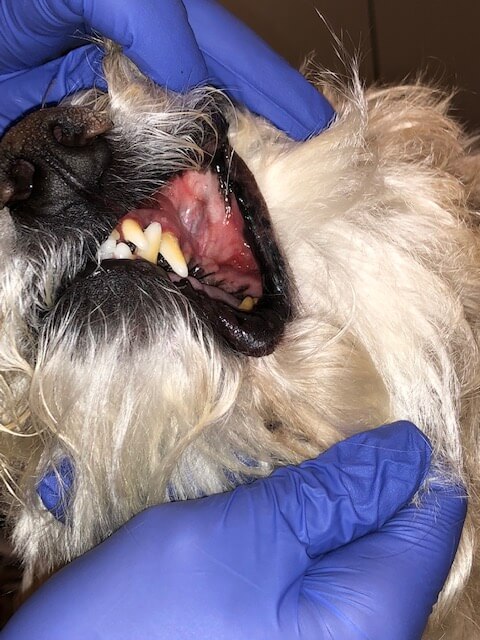
Oral squamous cell carcinoma treated with chemotherapy (gemcitabine), demonstrating strong response.
As melanomas have proven resistant to chemotherapy, there has been an effort to find a valuable systemic treatment to extend the prognosis in dogs beyond treatment achieved with local therapy alone. The melanoma vaccine was licensed by the FDA in 2010. The original work was unable to provide a median survival time as patients were alive at the end of the study period. It is important to note that this group of patients had locoregional disease control and were Stage II or III.11. A more recent study showed a MST of 455 days, for dogs with Stage I-III disease treated with locoregional control followed by the vaccine. A small subset of dogs had measurable disease and did show a response to the vaccine. More importantly, the vaccine provided an improved prognosis in dogs with Stage IV OMM.12
Oral tumors in dogs can be found incidentally or in the advanced stage. It is important to determine the specific tumor type, complete recommended staging, and provide clients with clear options regarding treatment in order to achieve the best outcome for their dog. The treatment recommendations are guided by the data, but also how best to treat an individual patient as part of their family.
For more information, please reach out to Dr. McQuown and MedVet Dallas’ Oncology team at 972.994.9110.
References
- JAVMA 2003. Williams LE et al. Association between lymph node size and metastasis in dogs with oral malignant melanoma: 100 cases (1987-2001).
- Vet Comp Oncol 2017. Skinner OT et al. Patterns of lymph node metastasis following bilateral mandibular and medial retropharyngeal lymphadenectomy in 31 dogs with malignancies of the head
- JAVMA 2019. Grimes JA et al. Histologic evaluation of mandibular and medial retropharyngeal lymph nodes during staging of oral malignant melanoma and squamous cell carcinoma in dogs.
- Vet Pathol. 2011. Smedley RC et al. Immunohistochemical diagnosis of canine oral amelanotic melanocytic neoplasms.
- Vet Rad Ultrasound 2003. Proulx DR et al. A retrospective analysis of 140 dogs with oral melanoma treated with external beam radiation.
- JAVMA 1984. Thrall DE. Orthovoltage radiotherapy of acanthomatous epulides in 39 dogs.
- JAVMA 2001. Rassnick KM et al. Use of carboplatin for treatment of dogs with malignant melanoma:27 cases (1989-200).
- JAVMA 2004. Boria PA et al. Evaluation of cisplatin combined with piroxicam for the treatment of oral malignant melanoma and oral squamous cell carcinoma in dogs.
- JAAHA 1998. Yoshida K et al. The effect of intralesional bleomycin on canine acanthomatous epulis.
- Vet Comp Oncol 2010. Kelly JM et al. Acanthomatous ameloblastoma in dogs treated with intralesional bleomycin.
- Am J Vet Res 2011. Grosenbaugh DA et al. Safety and efficacy of a xenogeneic DNA vaccine encoding for human tyrosinase as adjunctive treatment for oral malignant melanoma in dogs following surgical excision of the primary tumor.
- J Small Anim Pract 2017. Verganti S et al. Use of Oncept melanoma vaccine in 69 canine oral malignant melomas in the UK.