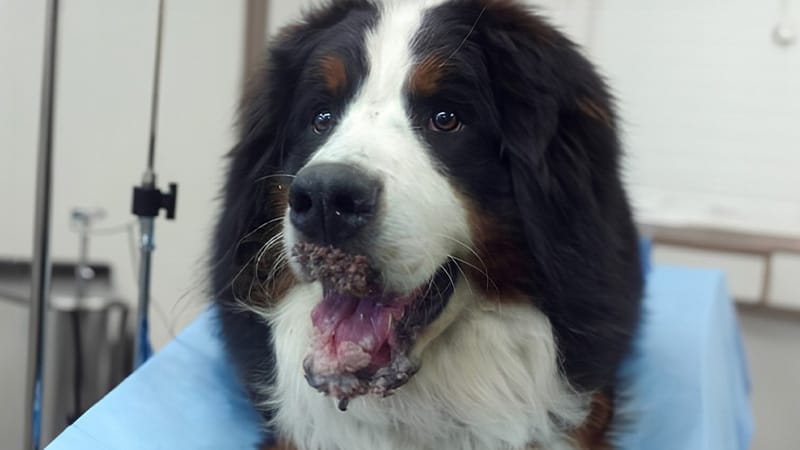
A 9-month-old, male Bernese Mountain dog was referred to dermatology services with a four-month history of canine oral papillomatosis involving the upper and lower lips, rostral aspect of the dorsal tongue, the hard palate and oral mucosae. Lesions had progressed rapidly over the four months prior to presentation despite earlier excision of a solitary lesion and a trial of azithromycin. Malodor and secondary infection were significant and affecting the dogs quality of life and interactions with family members. Additionally, due to the presence of papilloma lesions, he was restricted from social interaction with other dogs.
Upon examination, there were numerous intra-oral mass lesions multi-focally located on the hard palate, buccal mucosa, tongue, and in and around the teeth. On the rostral mandible, ventral to the nasal planum and on the rostral mandible at the mucocutaneous junction, there were large, necrotic, malodorous mass lesions adhering to underlying epithelial tissues by large stalks. No other body sites were affected and his general physical examination was normal.
Surgery
Given the extent of lesions, impaired quality of life and increasing interference with eating and drinking activities, surgical excision was recommended. Pre-anesthetic serum biochemistry profile and complete blood count did not reveal any abnormalities. On the day of surgery, the dog was anesthetized routinely. All intraoral lesions were removed with cauterization. The tongue lesions were excised with scissors and mucosal edges were sutured with vicryl ligature. The rostral mandible lesions were large, but rooted with “stalks.” They were isolated excised and the resultant mucocutaneous defect was sutured with PDS ligature. The dog was monitored overnight during anesthetic recovery without complication. The dog was discharged the following day with owner instructions to administer 100 mg tramadol orally every 12 hours for two days, 340 mg Baytril orally every 24 hours for three days, and 1000 mg cephalexin orally every 12 hours for three days. Sutures were removed after 14 days.
Histopathologic Examination
Excised specimens were submitted for histopathologic examination. Additional samples were retained in saline or frozen in the event we proceeded with potential viral isolation. The microscopic description of histologic specimens was “masses are composed of multiple papillary projections of heavily keratinized and markedly hyperplastic fronds of squamous epithelium supported by collagenous cores. Occasional enlarged and individualized squamous cells with glassy grey cytoplasm (koilocytes) are observed, as well as scattered intranuclear viral inclusion bodies. Neoplastic squamous cells are polygonal with prominent intercellular bridges, abundant acidophilic cytoplasm, round to oval nuclei with finely stippled chromatin, and one to multiple prominent nucleoli. Mitotic figures are occasionally observed, predominantly in the basal layer. Inflammation consists of stromal lymphocytes, plasma cells, and neutrophils. The specimens were consistent with viral papillomas.” (Alba Maria M. Shank DVM, MS, DACVP)

Post-Surgery
Upon reexamination 14 days post-operatively, all incisions had healed, and there was no evidence of new lesions. The owner reported that the pet had immediately returned to normal activity, was able to eat and drink without difficulty, and was once again an active member of family life. Three months after surgery, the dog continues to be lesion free.
Canine papilloma virus is characterized by benign tumors induced by infection of epithelial cells by species specific DNA papillomaviruses. At least 15 types of canine papillomavirus have been identified. The clinical manifestation of infection is dependent on the type of canine papilloma virus infecting the host. Canine oral papilloma virus (COPV, CPV1) causes oral papillomatosis. COPV is transmitted by direct and indirect contact with an incubation period of 1-2 months. Infection occurs at a site of damaged skin or mucous membranes. In general, COPV is usually self-limiting and will resolve in 3 months. Development of cell-mediated immunity by the infected host is required for papilloma regression. Young dogs with exposure to large numbers of other dogs at dog shows, dog day care, or at dog parks are at increased risk for infection.
COPV lesions may involve the oral cavity, lips, nasal planum, conjunctiva, haired skin of the face, and cornea. Lesions begin as multiple smooth white papules or plaques and progress to cauliflower-like masses. Anecdotally, dogs treated with immunosuppressive doses of cyclosporine, glucocorticoid therapy or cancer chemotherapy have developed lesions in adulthood.
Diagnosis of papillomavirus is made via histopathology, history, signalment and clinical signs. Papilloma virus antigen may be detected by immunohistochemistry or PCR. The prognosis is usually good, as most COPV lesions spontaneously regress without intervention after development of a host cell-mediated immune response. Malignant transformation to SCC is possible but rare.
For treatment of COPV lesions that do not regress as expected, surgery may be curative for persistent solitary lesions. Care should be taken intra-operatively to avoid seeding the surgical site with viral particles. Cryotherapy and laser ablation are often effective, but may require several procedures. Use of interferon has been variably effective. Autogenous vaccines are of undocumented efficacy.
A new recombinant canine oral papillomavirus vaccine produced by Georgetown University Medical Center shows promise for treatment of refractory canine oral papillomas. The laboratory was involved in the production of Gardasil, the preventive vaccine for human papilloma virus (HPV). The vaccine consists of the major capsid protein L1 of the COPV. In one published report, oral papillomas regressed completely by the time the last injection of the COPV-L1 vaccine series was administered, and lesions had not recurred after 60 months of observation. One developer of this COPV-L1 vaccine, Hang Yuan, has subsequently treated 19 dogs with COPV L1 vaccine. In total, 31.6% had progression of lesions, 21% had stabilization of lesions and 47.4% had total regression of lesions without recurrence. Overall, 2/3 of the treated dogs responded. In those that did not respond, he speculates they had a specific immunodeficiency preventing adequate immune response or lesions that did not respond had already undergone malignant transformation to squamous cell carcinoma rendering the tissue not responsive to the antiviral vaccine . The most widely used application of this COPV L1 vaccine produced by his laboratory is for large dog groups where the risk of infection is high due to close cohabitation. The vaccine has resulted in prevention of COPV lesions in such groups.
In closing, what made this COPV case intriguing to me was the extraordinarily atypical biological behavior of this dog’s COPV and the significant distortion of the lesions to his physical appearance. In addition, the extent of the lesions impaired his quality of life and disrupted the development of the human-animal bond within his family. Ultimately, an immediate and aggressive surgical intervention quickly resolved the entire problem for the dog and his humans. While we were prepared to develop and administer the novel COPV-L1 vaccine to this patient, it was not required. However, we wanted to share this case and introduce this available treatment option. Finally, this case underscores the need for collaboration between veterinary specialties and our human colleagues on the front lines of research and development from time to time.